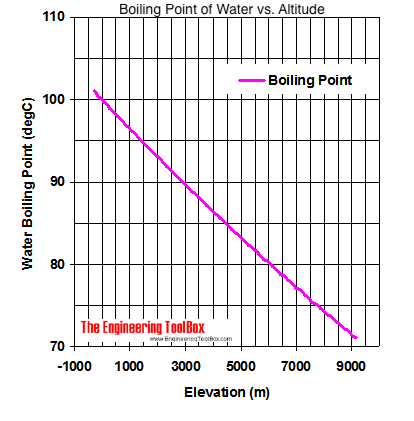
![SOLVED: Homcwork; Name: Althe top of Mount Everest, water boils at 70 %C. Explain why lhis 1s lower than its usua] boiling point of 1OOPC. 2. ) How much heat is needed SOLVED: Homcwork; Name: Althe top of Mount Everest, water boils at 70 %C. Explain why lhis 1s lower than its usua] boiling point of 1OOPC. 2. ) How much heat is needed](https://cdn.numerade.com/ask_images/4cc58932dc364e578898e564645cccc7.jpg)
SOLVED: Homcwork; Name: Althe top of Mount Everest, water boils at 70 %C. Explain why lhis 1s lower than its usua] boiling point of 1OOPC. 2. ) How much heat is needed

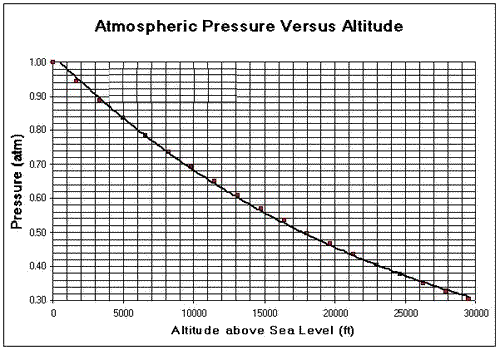
Universal science facts - Why does water boil at 71 degrees on Everest? Liquids start boiling when their vapour pressure equals atmospheric pressur. Atmospheric pressure go on reducing as we go higher

TVO Learn Mathify on Twitter: ""Q: What do you call a teapot of boiling water on top of Mount Everest? A: A "high-pot-in-use!" #mathjoke" https://t.co/Lo2EFY9Bed" / Twitter