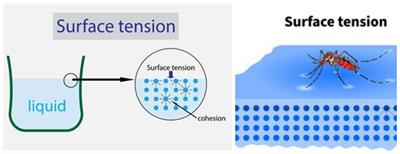
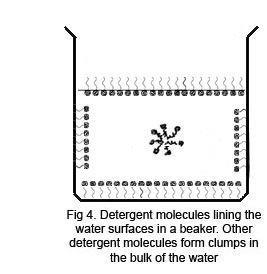
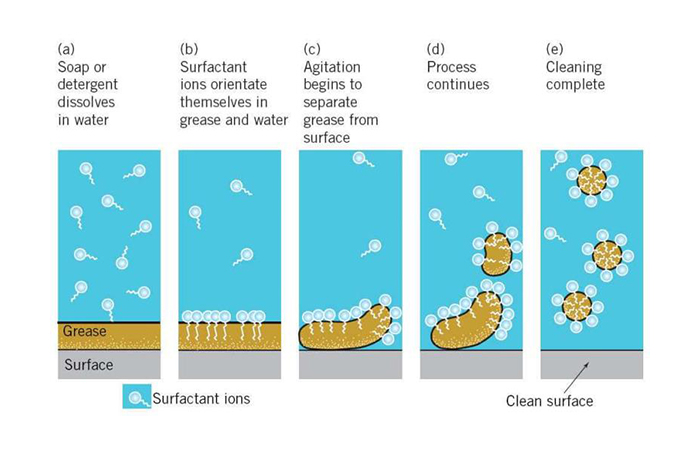
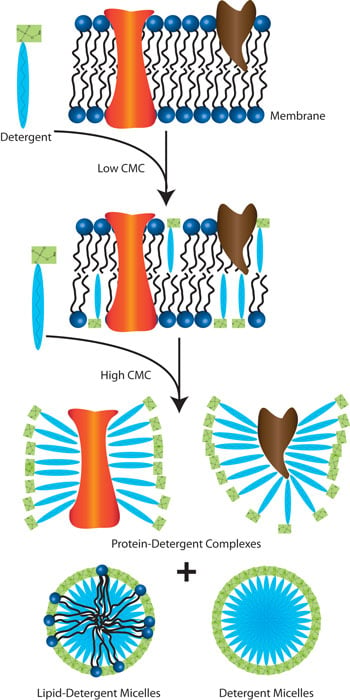
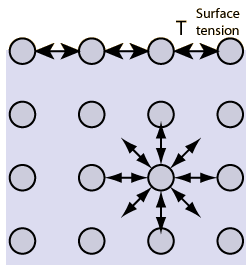
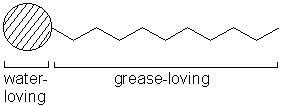Explain why (a) The angle of contact of mercury with glass is obtuse, while that of water with glass is acute. (b) Water on a clean glass surface tends to spread out
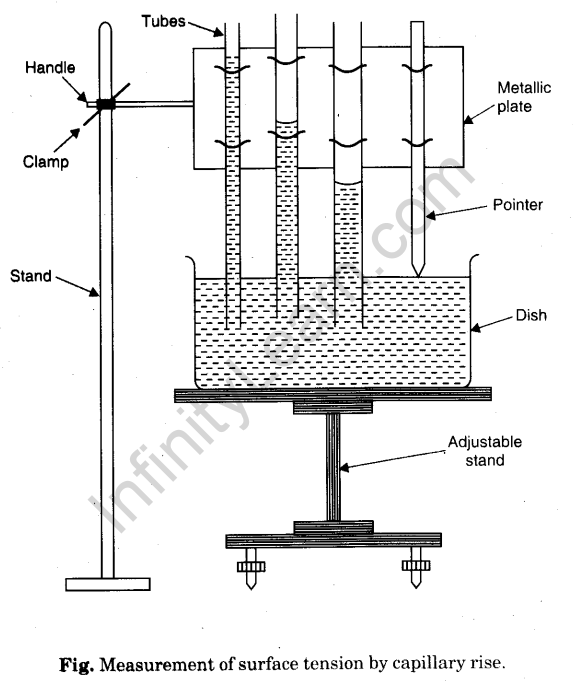
To Study the Effect of Detergent on Surface Tension of Water by Observing Capillary Rise - Infinity Learn

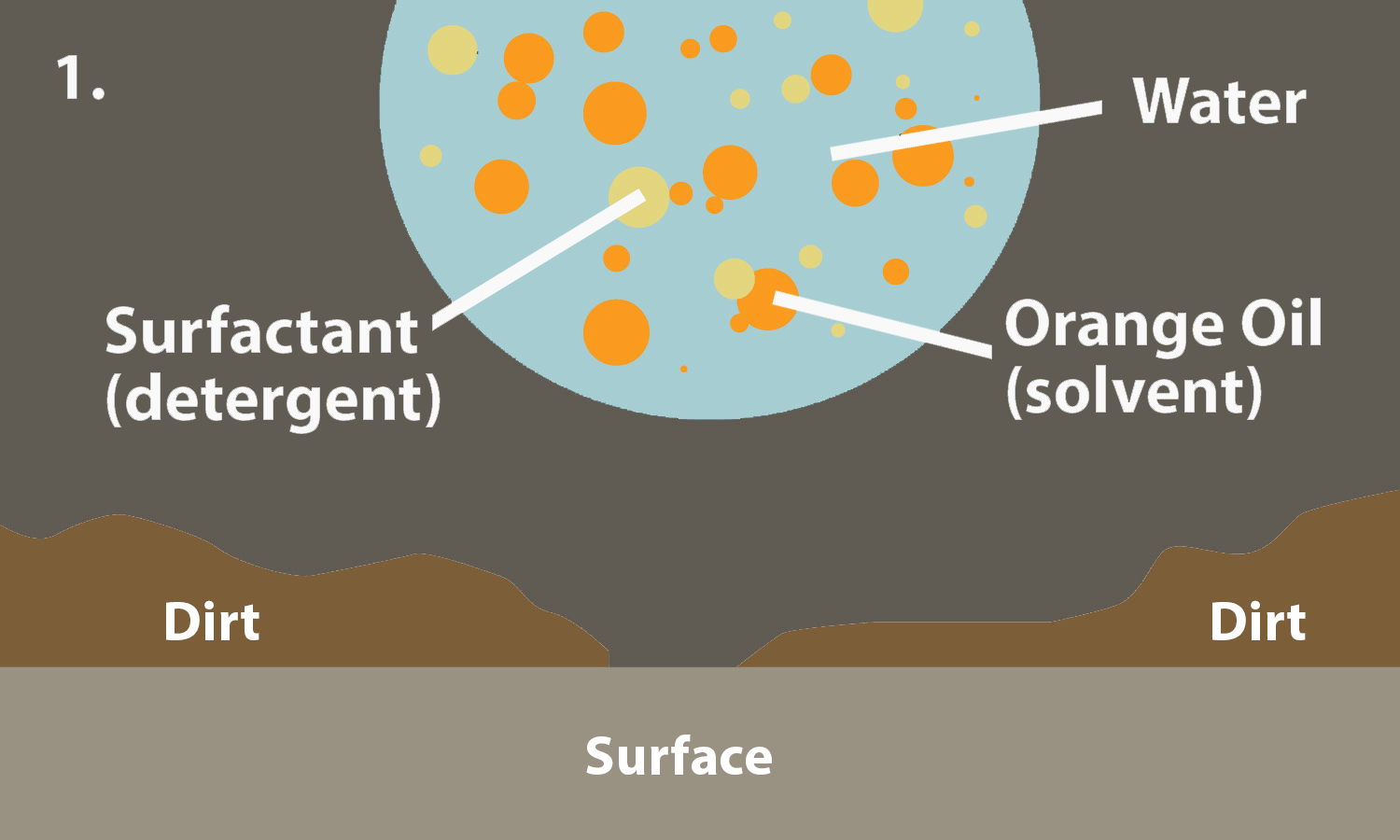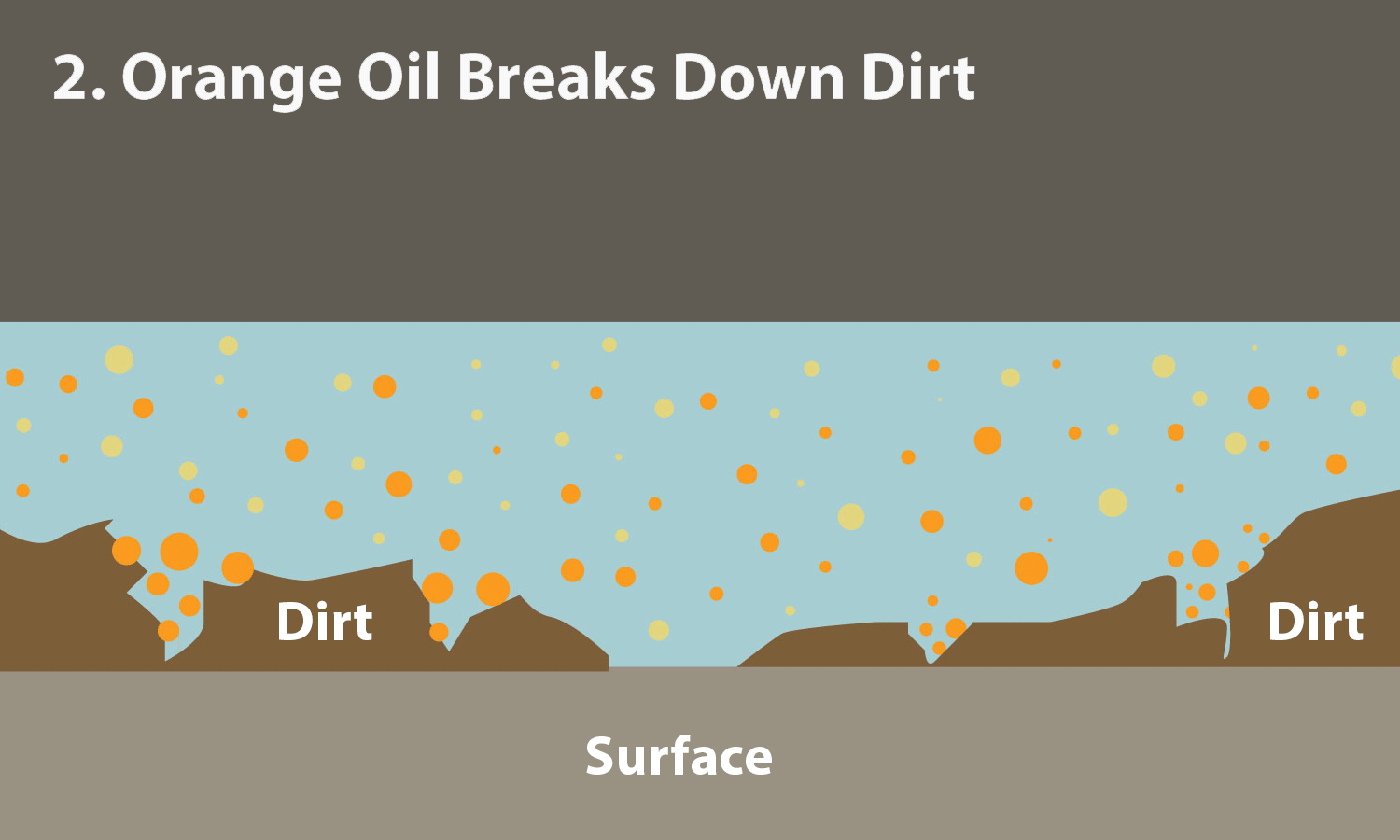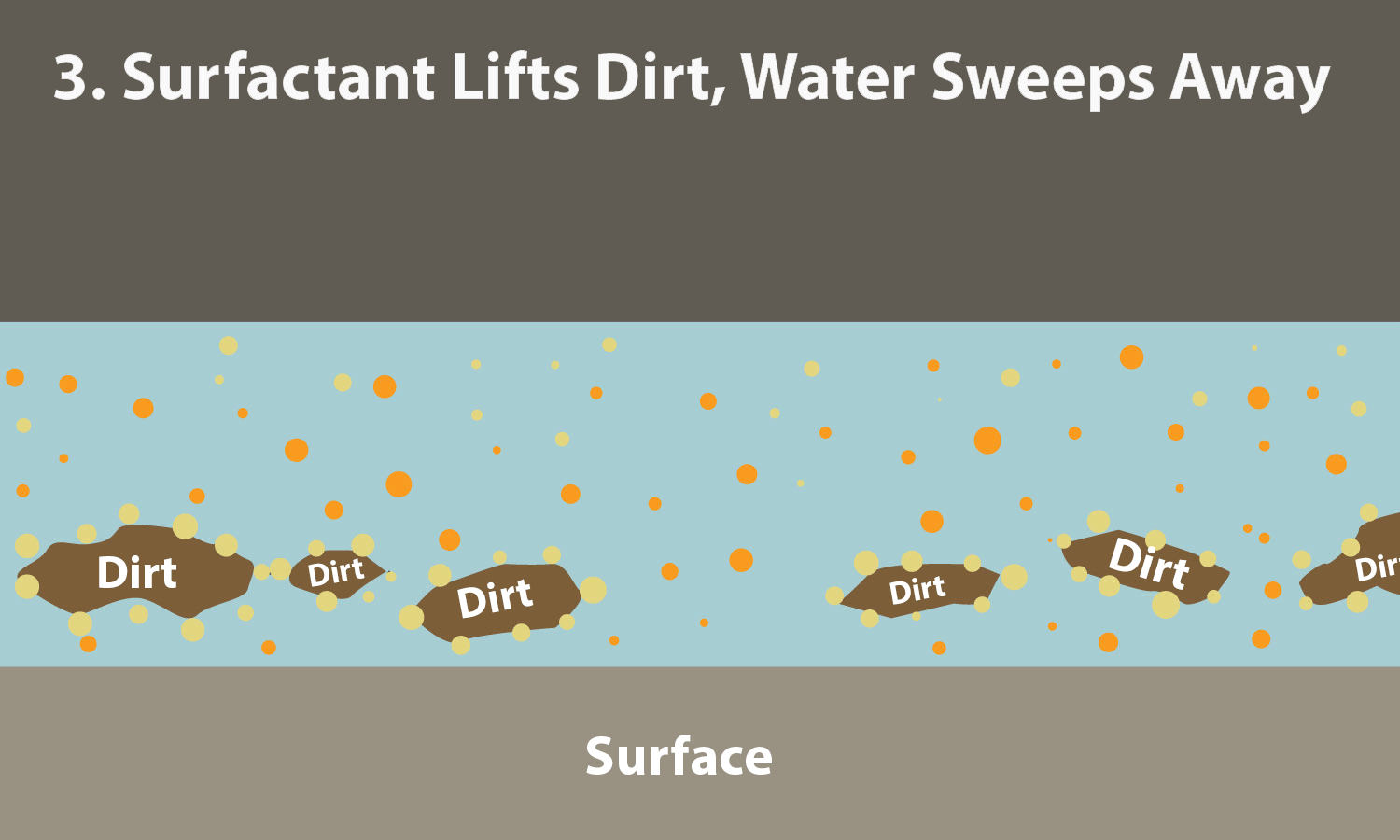
Phase-Dependent Surfactant Transport on the Microscale: Interfacial Tension and Droplet Coalescence | Langmuir